Web Content Display Web Content Display
Authigenic carbonates
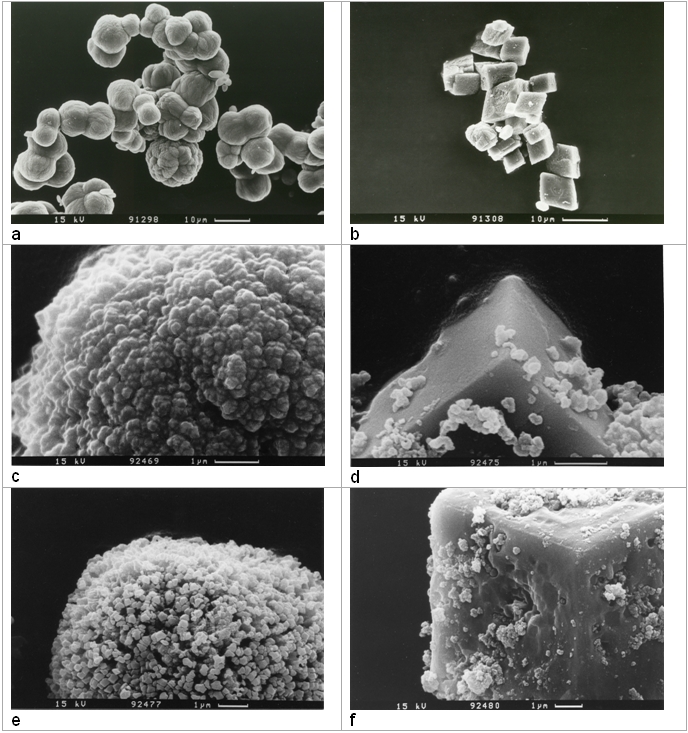
Authigenic carbonate formation related to mud volcanism and cold seeps is a widely observed process at active continental margins. It is also a frequent phenomenon along the Middle American Subduction Zone. High Mg-calcite (HMC) concretions were determined in surface sediments along the Costa Rica forearc. HMC is most abundant at water depth between 1000 and 2000m. To calculate fluid composition and temperature conditions during formation the stable oxygen isotope fractionation behaviour of HMC-H2O have to be known precisely. During SFB574-B5 the oxygen isotope fractionation equation of the HMC-water system was investigated experimentally by temperature controlled carbonate precipitation experiments. The experiments were conducted at temperatures between 25 and 80°C from isotopically defined solutions. HMC with MgCO3-contents between 6 and 32% formed from mixtures of CaCl2-MgCl2 and Na2CO3 solutions. The degree of the Mg2+-incoporation into the high magnesium calcite crystal lattice could be correlated to the saturation degree of magnesite in the initial aqueous mixture. Our data show that this relation is valid between 25 and 80°C. Although mostly microcrystalline rhombohedral HMC (size: ~300 nm-10 µm) was formed during our experiments amorphous carbonate co-precipitating occurred (e.g. Fig. 1a). Nearly quantitative dissolution of amorphous carbonate was achieved by applying a partial acid digestion method (compare Fig. 1c and e). This HMC purification process did not change the general structural order or the oxygen isotopic composition, however, etching and small acid pits were observed at high magnification in SEM (Fig. 1f).
where x is 103/T in Kelvin and CMg is the percentage of Mg2+ incorporated in the crystal lattice.
Fig. 1: Scanning electron micrographs of high Mg-calcite crystals precipitated at 25°C (a,c, e) and 80°C (b,d, f). Micrographs e and f show the crystal morphology after applying a partial acid digestion method to remove amorphous carbonate. Further readingChacko T. and Deines P. (2008) Theoretical calculation of oxygen isotope fractionation factors in carbonate systems. Geochim. Cosmochim. Acta 72, 3642-3660. |
 Events Events
Kieler Wissenschaftler fühlen den 'Puls der Erde' Wie funktioniert die Recyclingmaschine der Erde?Nach elf Jahren endet der Kieler Sonderforschungsbereich 574 zu Subduktionszonen Final colloquium of SFB 574 Teilprojekt ÖffentlichkeitsarbeitMEERESFORSCHUNG FÜR MICH UND DICH |
|
©SFB574 // Wischhofstrasse 1-3 // D-24148 Kiel // T. +49 (0)431 600 1413 // elange [AT] geomar.de