 Web Content Display Web Content Display
Methods
The fate of methane emitted from vent sites is monitored from the sediment through the water column to the ocean surface waters. Methods include in-situ measurements in benthic chambers deployed by benthic landers and/or ROV as well as video-guided multicorer deployments at the sediment/water interface and profiling CTD/Rosette water sampling.
Due to the ROV Kiel 6000 we explore the seabed with acoustic (sonar) and optic (video) methods. By using the two manipulators push cores, a pore water sampler and a temperature lance can be exactly positioned for sampling which enables in combination with the large area survey regionalizing of the data.
Fluid flow is measured in situ by the employment of benthic chambers, positioned either by ROV or video-guided landers at the seafloor. These chambers are driven slowly by motors into the sediment and obtain in definite time intervals water samples from the overlying water of the chamber. After the end of the incubation the sediment will be retrieved and the samples can be, analyzed on board for the chemical components (e.g. oxygen, methane, sulfide, sulfate). From their change over time the turnover rate and by using numerical modeling fluid flow is calculated.
In the Lander BIGO (BioGeochemical Observatory) 2 chambers can be incubated simultaneously and the oxygen concentration can be controlled by a gas exchange system.
Fig. 4: Deployment of 2 benthic chambers in BIGO, gas exchange system on the right side. In the Profiler Lander microelectrodes are deployed for the in situ measurement of oxygen, sufide and pH. At the same time the current regime of the bottom water will be monitored by up and down looking acoustic current profilers (ADCPs).
Fig. 5: Profiler Lander with ADCPs (white circles) und profiler, right: profiler with microelectrodes. In the lab micro electrodes are applied to monitor high-resolution profiles of oxygen, sulfide, pH, and redox potential in surface sediments. This technique enables the characterization of microbial micro niches and to identify the activity of anaerobic oxidation of methane, sulfate reduction and sulfide oxidation. The CTD/Rosette measures conductivity, temperature and pressure in the whole water column. Additionally, up to 24 water samples can be obtained from different water depth, which are analysed in the lab. At the same time the CTD can be equipped with additional sensors (e.g. oxygen, methane, turbidity) to detect and sample methane anomalies in the water column. The water samples will be degassed and the extracted gas will be injected onboard in a gas chromatograph (GC) for hydrocarbon analyses. The dissolved gases from the sediments will be extracted as well and the ratio of methane to higher hydrocarbons determined in the pore waters and the water column over the seep areas.
Radioactive tracers (14CH4 and 35SO42-) are used to obtain the turnover rates of AOM and SR. For that purpose the sediment cores will be subsampled vertically with pushcores. The radiotracers will be injected at 1 cm intervals and incubated for 24h by the in-situ temperature of the sediment, anoxic and in darkness. After the incubation time the bioactivity is stopped by zinc acetate or sodium hydroxide. The concentrations/activity of the radiotracers - the educts and products of the reactions - is measured in a scintillation counter. By this it is possible to compare the tracer concentration to the whole concentration and calculate the turnover rate.
Catalyzed reporter deposition fluorescence in vitro hybridization (CARD-FISH) is used to identify and quantify specific microbial groups in environmental sediment. There for the hole sample are fixed and the cells permeabilized, before the hybridization accomplishes. The oligonucleotide probe consists of horseradish peroxidase and a base sequence which is connecting the rRNA of the microbial group. After a signal amplification, by tyramid, it is possible to microscope and quantifies the cell density. To localize the syntrophic partner a double-hybridization is possible. Controlled is this technique by the DAPI-method, which stains the DNA of all cells in the sample.
Fig. 9: Picture of colored ANME-2, left due Cy-3 and right due DAPI To understand the control mechanisms and dynamics of the benthic methane filter, we use sediment flow through systems. The system includes reservoirs for two different seawater media. The first, from above, contains sulfate and the second, from below, methane, which is enriched due a methane headspace. Both media are kept anoxic due Nitrogen. Pumps carry this media in the sediment column which is located in an incubator. The outlet is located on the top, so that the methane-medium is flowing advectively through the core, whereas the sulfate-medium penetrates only due diffusion.
Due to different in vitro experiments, the sediment-microbe interactions and their response times are investigated. Hereby, abiotic factors such as pH, temperature, alkalinity, advective flow as well as concentration of methane, sulfate, and sulfide will be varied. Sampling holes in the core liners enable an intensive study of these parameters as well as the abundance and diversity of microorganismens in different sediment depths. These parameters will be determined by micro sensors, pore water analysis and CARD-FISH. Data gained by the above methods will be integrated into numerical models to assess the impact of transport processes on biomass distributions, rates of anaerobic oxidation of methane (AOM) and methane releases from the seafloor. In summary, our approach will combine different modern methods including in-situ, ex-situ, and in-vitro techniques as well as numeric modeling to investigate the fate of methane from different perspectives. By comparison of the obtained information with measurements from other margin settings gaps in the global methane budget will be closed.
|
 Events Events
Kieler Wissenschaftler fühlen den 'Puls der Erde' Wie funktioniert die Recyclingmaschine der Erde?Nach elf Jahren endet der Kieler Sonderforschungsbereich 574 zu Subduktionszonen Final colloquium of SFB 574 Teilprojekt ÖffentlichkeitsarbeitMEERESFORSCHUNG FÜR MICH UND DICH |
|||||||
©SFB574 // Wischhofstrasse 1-3 // D-24148 Kiel // T. +49 (0)431 600 1413 // elange [AT] geomar.de






 Fig. 1: Recovery of the video-guided multicorer.
Fig. 1: Recovery of the video-guided multicorer.
 Fig. 3: Deployment of a benthic chamber by ROV
Fig. 3: Deployment of a benthic chamber by ROV



 Fig. 6: Deployment of the CTD/Rosette
Fig. 6: Deployment of the CTD/Rosette Fig. 7: The GC-C-irMS for measurement of δ13C, on top the GC and below the mass spectrometer.
Fig. 7: The GC-C-irMS for measurement of δ13C, on top the GC and below the mass spectrometer. Fig. 8: Determine of sulfate reduction
Fig. 8: Determine of sulfate reduction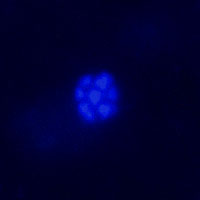
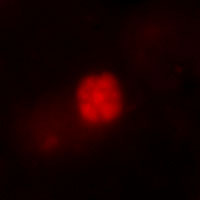
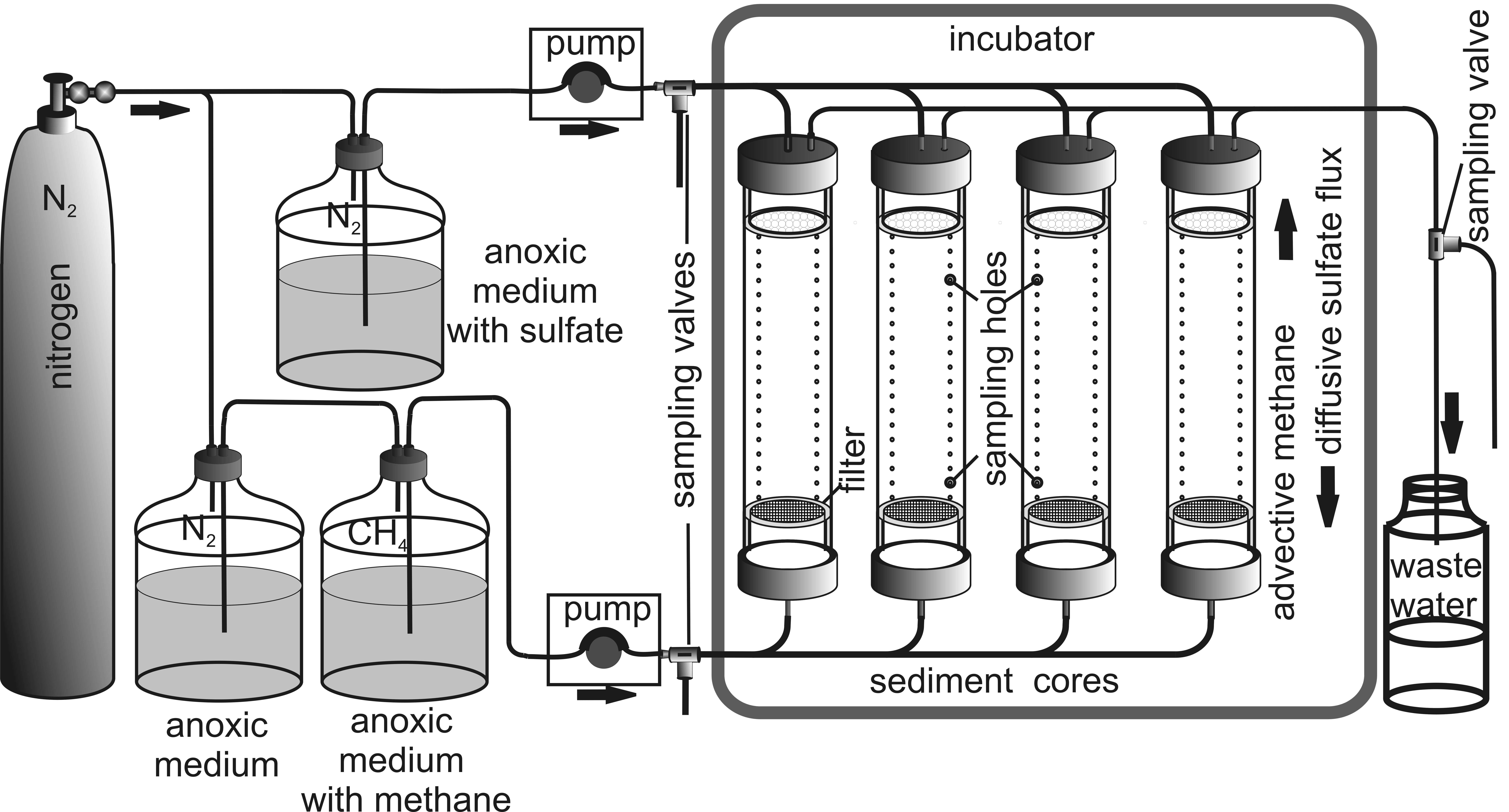 Fig. 10: schematic picture of the Sediment flow through system
Fig. 10: schematic picture of the Sediment flow through system